From:Guest Contributor
A joint research team at Tohoku University and the University of California, Los Angeles (UCLA) has made significant progress in high-voltage metal-free lithium-ion batteries using small organic molecules, croconic acids. This breakthrough brings us one step closer to realizing metal-free, high-energy and inexpensive lithium-ion batteries.
Unlike traditional lithium-ion batteries that rely on rare earth materials such as cobalt and lithium, organic batteries use naturally abundant elements such as carbon, hydrogen, nitrogen and oxygen. In addition, organic batteries use organic materials to make them lightweight, so it has a larger theoretical capacity than conventional lithium-ion batteries. However, most of the organic batteries reported so far have relatively low (1–3 V) operating voltages. Increasing the voltage of organic batteries will lead to higher energy density batteries.
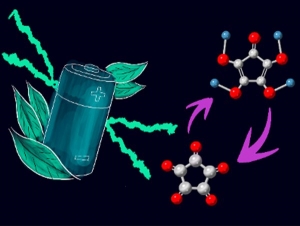
Itaru Honma, a professor of chemistry at Tohoku University’s Institute of Multidisciplinary Research for Advanced Materials, Hiroaki Kobayashi, an assistant professor of chemistry at Tohoku University, and Yuto Katsuyama, a graduate student at UCLA, found that croconic acid, when used as a lithium-ion battery cathode material, maintains a strong working voltage of around 4 V.
Croconic acid has five carbon atoms bonded to each other in a pentagonal form, and each of the carbons is bonded to oxygen. It also has a high theoretical capacity of 638.6 mAh/g, which is much higher than the conventional lithium-ion battery cathode materials (LiCoO2 ~ 140 mAh/g). “We investigated the electrochemical behavior of croconic acid in the high-voltage range above 3 V using theoretical calculations and electrochemical experiments,” said Kobayashi. “We discovered that croconic acid stores lithium ions at roughly 4 V, giving a very high theoretical energy density of 1949 Wh/kg, which is larger than most inorganic and organic lithium-ion batteries.”
Although the study did not reach the theoretical capacity, the researchers are optimistic that this can be improved by developing stable electrolytes and chemical modification of croconic acid at high pressures. Since most electrolytes cannot withstand such strong ketoacid working voltages, the development of new electrolytes is crucial.
Learn more: https://cleantechnica.com/